ACRUX TIMELINE

Launch of 90g Dapsone 5%, Gel

FDA approval and launch Nitroglycerine 0.4%, Ointment

FDA Approval of Dapsone, 7.5% Gel

Launch Dapsone 5%, Gel.

As at August 2023, Acrux has six approved products with 3 ANDA's under review by the FDA.

AB-rated generic version of Emla® Cream (Lidocaine 2.5% and Prilocaine 2.5% Cream) launched in USA Dec 2022 by our Acrux licensee, Padagis.
Acrux has had 6 ANDAs accepted for review by the FDA.

Development agreement with commercial organisations for marketing of the Acrux developed ANDA products in the United States.

Submitted third ANDA (generic) product to the FDA for review.
Lenzetto® marketed in over 30 countries in Europe and other countries outside Europe.
Active pipeline of 14 generic products.

Submitted first two ANDA (generic) products to the FDA for review

Lenzetto® launched in Europe by our licensee Gedeon Richter.

Evamist® licensed to Perrigo in US
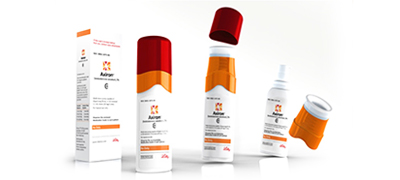
Axiron® launched by Eli Lilly

Acrux submits Axiron® NDA to FDA

Evamist® launched by KV Pharmaceutical

Evamist® licensed to KV Pharmaceutical in US

Acrux listed on ASX (ACR:ASX)
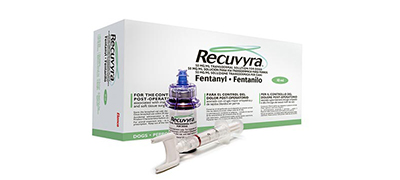
Recuvyra® Veterinary product licensed to Elanco globally.

First US patent granted

Acrux incorporated
